
China team develops new high-performance cathode for Li-sulfur batteries
Researchers led by Prof. Jian Liu and Prof.Zhongshuai Wu from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences have developed Fe1-xS-decorated mesoporous carbon spheres as a cathode material for lithium-sulfur batteries.
Fe1−xS‐NC has a high specific surface area (627 m2 g−1), large pore volume (0.41 cm3 g−1), and enhanced adsorption and electrocatalytic transition toward lithium polysulfides (LiPs). The material shows excellent polysulfide catalytic activity and cyclic stability. An open-access paper on their work is published in the journal Advanced Energy Materials.
Lithium-sulfur batteries have a high theoretical energy density of 2600 Wh kg-1 and a theoretical capacity of 1675 mAh g-1. However, the slow conversion reaction dynamics of sulfur in the process of charging and discharging lead to low utilization rate of sulfur and a serious polysulfide shuttle effect. This further causes low capacity and stability of lithium-sulfur batteries.
An electrocatalytic system is needed such that the catalytic transformation of polysulfide can be realized efficiently and steadily under high sulfur loading, resulting in high cyclic stability of the lithium-sulfur battery.
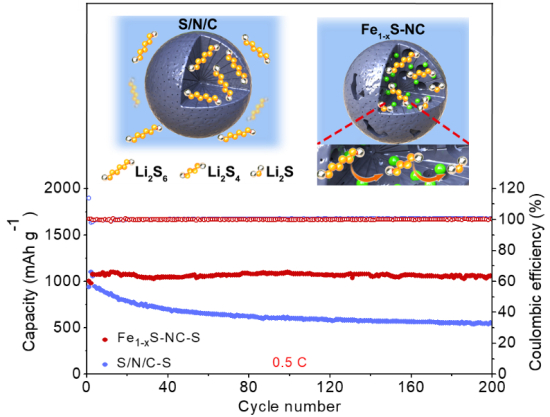
In the current study, the researchers designed a mesoporous carbon material decorated with highly dispersed Fe1-xS electrocatalyst nanoparticles (Fe1-xS-NC), and applied it as lithium-sulfur battery cathode for high catalytic activity and high sulfur loading.
The material features low mass density, high porosity, and highly dispersed electrocatalyst, which significantly improves the adsorption and catalytic conversion capacity of polysulfides.
The researchers found that there was virtually no decay in capacity of Fe1-xS-NC from an initial value of 1070 mAh g-1 after 200 cycles and under a current density of 0.5 C.
“The high electrocatalytic activity of Fe1−xS nanoparticles coupled with their high electronic conductivity and accessibility allow for efficient adsorption and conversion of LiPs to Li2S. The electrocatalysts are well dispersed in the hierarchically porous carbon spheres with large mesoporous cavities, which can accommodate high sulfur loading, modulate volume variations during charge/discharge cycles and enhance mass transfer of electrolyte to active sites. As a result, high‐performance cathode with exceptional stability (no capacity fading at 0.5 C after 200 cycles), high rate capability and excellent cycling performance at high sulfur loading of 8.14 mg cm−2, are achieved for LSBs. This work demonstrates the potential applicability of well-dispersed metal sulfides at low metal loading inside highly porous carbon spheres as high‐performance nanoreactors for LSBs. The results can also open opportunities for construction of more complex architectures such as hollow structures, yolk–shell or multishell particles doped with metal sulfides via molecular‐level design to improve the performance of LSBs in terms of long‐term cycling stability and high sulfur loading. Molecular level design could also be an attractive method for the synthesis of highly dispersed metal nitrides, oxides, phosphides, or halides inside a carbon support by the suitable choice of a precursor and synthesis methods.” (Boyjoo et al.)







Recent Comments